Researchers have reported on an innovative interfacial engineering approach to enable fast charge-discharge rates in solid-state lithium metal batteries. They have found that nanoscopic refractory metal layers like Tungsten could improve the performance of these batteries which are crucial for purposes like electrical mobility.
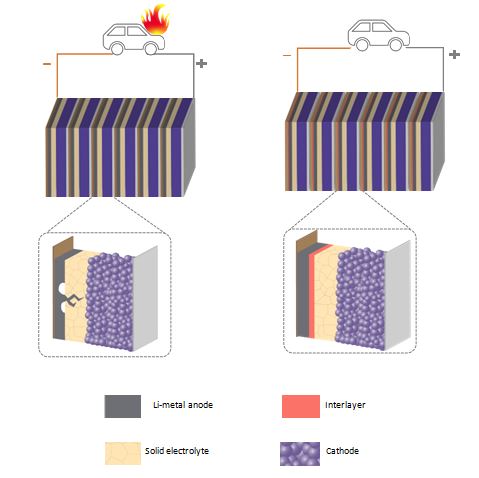
Conventional Li-ion batteries employ a graphite anode, a liquid electrolyte, and a transition metal cathode. However, the liquid electrolytes are flammable and degrade at high temperatures leading to poor battery life and in extreme cases lead to battery fires. Replacing the liquid electrolyte in a conventional Li-ion battery with a ceramic solid electrolyte and simultaneously replacing the graphite anode with a metallic lithium anode could enable safer Li-ion batteries that also last long on a single charge.
However, a long-standing challenge with solid state batteries is the growth of lithium dendrites that short circuits the cells and this is accentuated during fast charging.
Based on extensive fundamental electrochemical measurements performed over several hundreds of solid-state half cells and subsequent nano-characterization, researchers from the Indian Institute of Science (IISc) realized that dendrite growth was a manifestation of a deeper fundamental process: diffusive growth of lithium voids that are forming during discharge. The researchers identified that the growth of lithium voids during discharge leads to dendrite growth during charge.
The team consisting of Vikalp Raj, Victor Venturi, Varun R Kankanallu, Bibhatsu Kuiri, Venkatasubramanian Viswanathan and Naga Phani B Aetukuri found that at the edges of the microscopic voids, Li-ion currents are concentrated. The currents at these edges are around 10000 times larger than average currents in the cell. Hence it is necessary to impede void growth to prevent dendrite growth.
Experimenting with an ultrathin layer of refractory metals between the lithium anode and the solid electrolyte, the researchers noted that tungsten is an ideal candidate to impede lithium vacancy motion due to its low solubility for lithium and therefore delay void growth. They collaborated with researchers from Carnegie Mellon University to corroborate their work through computational methods.
Facilities created under the Materials for Energy Conservation and Storage Platform (DST-MECSP) program, Technology Mission Division (Energy, Water and Others), Department of Science and Technology (DST) under the DST-IISc Energy Storage Platform on Supercapacitors and Power Dense Devices has been pivotal in taking the work forward. The work is published in Nature Materials.
The researchers acknowledged the support from to Dr. Ranjith Krishna Pai, Scientist ‘E’ (TMD-EWO), DST and (Late) Shri. Dr. Sanjay Bajpai, Former Head (TMD-EWO), DST for conceptualizing this program which brought several essential facilities for energy research under a single roof and Dr. Anita Gupta, Head (TMD-EWO), DST, for extending it.
The team now intends to build on this advance to develop full solid-state cells that could enable charging in less than an hour, offer up to 1000 or more cycles while withstanding high temperatures of 45 ºC or higher outcompeting conventional Li-ion cells at a cost that is at par or lower than the cost of conventional Li-ion cells.
Link to publication:
https://www.nature.com/articles/s41563-022-01264-8
Figure 1: A liquid electrolyte-based Li-ion battery with a lithium anode (left) could be susceptible to battery fires. By contrast, the solid-state Li-ion battery with a lithium anode is safe and charge faster with the lithium insoluble interlayer approach proposed by the researchers

Figure 2: Battery and supercapacitor test facilities at the DST-IISc Energy Storage Platform for Supercapacitors and Power Dense Devices
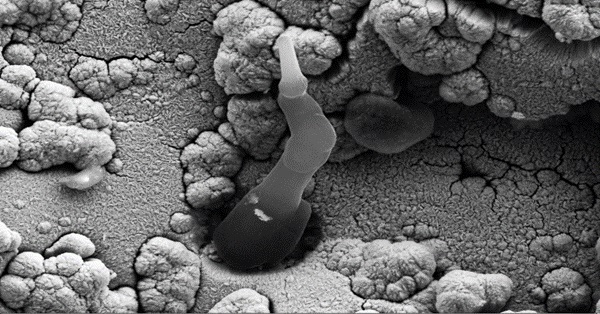
Figure 3: Scanning electron microscopy image of a lithium filament
SNC/RR